 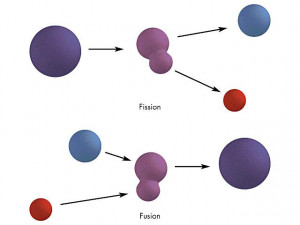
Young University of Illinois at Chicago via The process by which nitrogen is converted to carbon-14 is an example of neutron capture, in which particles are absorbed by the nucleus of another atom to form a new element. Albert Einstein, himself one of these refugees, was persuaded to write a letter to President Franklin Roosevelt in August 1939 in which the alarming possibilities were outlined. 11.6: Nuclear Fission Last updated 11.5: Radioactive Half-Life 11.7: Nuclear Fusion Paul R. Examples of nuclear fission processes are illustrated. A small amount of mass is 'left over' and released as energy. The nucleus splits into two isotopes, each of which is roughly half the mass of the original atom. A neutron (generally produced by some controlled process, not usually a natural event) collides. Nuclear fission is the process of a neutron colliding with a nucleus. Fusion, in contrast, occurs when two or more smaller atoms fuse together, creating a. The possibility that Nazi Germany might develop such a bomb and use it did not seem remote, especially to those scientists who had recently fled Nazi and Fascist Europe and come to the United States. The example above illustrates the basic nuclear fission process. In fission, an atom is split into two or more smaller, lighter atoms. Because the sum of the mass numbers of the reactants must equal the sum of the mass numbers of the products: 25 + 4 A + 1, or A 28. Below is a simple fission process: Figure 1. For U 235 on average 2.5 neutrons are emitted, starting on average two more fission reactions. A tremendous amount of energy is produced by the fission of heavy. (b) The larger fission products of U-235 are typically one isotope with a mass number around 85105, and another isotope with a mass number that is about 50 larger, that is, about 130150. where A is the mass number and Z is the atomic number of the new nuclide, X. One of the most well-known and useful examples of a chain reaction is of U 235 which is used to harness nuclear energy. 2: (a) Nuclear fission of U-235 produces a range of fission products. Adolph Hitler had been in power in Germany for 6 years, and Europe was teetering on the brink of World War II. The nuclear reaction can be written as: 12 25 Mg + 2 4 He 1 1 H + Z A X. Any nuclear reaction in which the products have a higher mass defect/binding energy than the reactants will release energy. Inside the sun, fusion reactions take place at very high temperatures and enormous gravitational pressures. Note: This is about the same quantity of heat energy as that produced by burning 3 tons of bituminous coal!Ĭalculations similar to that just performed soon persuaded scientists in 1939 that the fission of uranium was highly exothermic and could possibly be used in a super bomb. Look up during the day to see one of the most powerful examples of a nuclear reactor: the sun. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
